

Project Solutions
Vehicle Battery Jogger-keep your battery fresh and young (4)
Published:2011/8/8 0:50:00 Author:Phyllis From:SeekIC
By Karel Walraven
FETs
We have listed several options for FET T1, but the choice here is not critical. It’s also unnecessary to make any changes to the circuit if you use it at 6 V. Still, in this case the preferred type for Tl is the IRF540. That’s because this type of FET works with a lower gate voltage than the FETs in the BUZ family. For the same reason, FETs designated as ’logic-level FETs’ are more suitable if you want to use the circuit at 6V, but they aren’t essential. In principle, you can thus use any desired type of n-channel power FET that can handle at least 20 A at 50 V.
You’re probably wondering how a FET rated at 20 A can handle a current of 40 A or more. To understand why this is possible, have a look at the ’Safe Operating Area’ chart for the BUZ10 (Figure 3). It shows how much current can flow continuously (20 A), and how much current can flow for a short time. As the FET only conducts for a few tens of microseconds each time, the maximum allowable current during this short interval is just under 100 A. This information con also be obtained from the ’Absolute Maximum Ratings’ in the dale sheet, which specify 23 A and 92 A respectively. 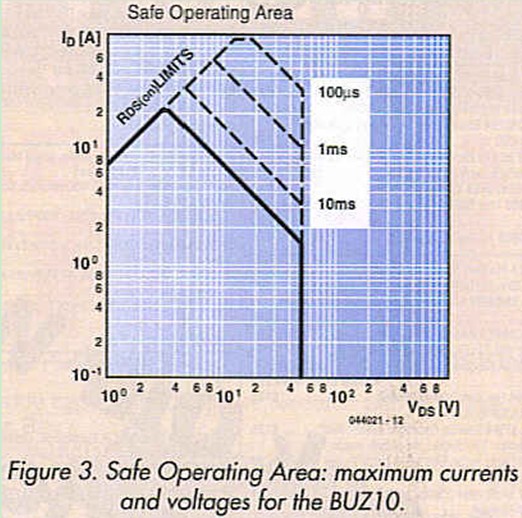
Sulphation
The capacity of a lead-acid battery gradually decreases during its service life due to normal wear and tear, for a variety of reasons. However, the aging process is considerably accelerated if an uncharged battery is stored for longer than a week at a temperature below 10 CC or remains unused (while charged) for an extended length of time. That’s because in such situations, a layer of non-conductive lead sulphate (PbSO-j) forms on and around the electrodes. This reduces the effective surface area of the electrodes, thus decreasing the capacity of the battery. This process is difficult or impossible to reverse by normal charging and discharging.
However, it is possible to break down sulphate crystals into lead and sulphuric acid by using high (or very high) charging currents. This method can be used to restore at least part of the lost capacity (see ’Lead-acid battery revitaliser’ in the September 2001 issue). Still, it’s better to prevent the formation of sulphate. One way to do this is to periodically apply a load to the idle battery. The circuit described in this article is especially suitable for this purpose.
Reprinted Url Of This Article: http://www.seekic.com/blog/project_solutions/2011/08/08/Vehicle_Battery_Jogger_keep_your_battery_fresh_and_young__(4).html
Print this Page | Comments | Reading(328)
Article Categories
New published articles
· Imagination works with TSMC to develop FinFET process
Author:Ecco Reading(30188)
· XMOS pushes event-driven MCUs with lower price
Author:Ecco Reading(3461)
· Intel brings upgraded 32-nm SoC for smartphones
Author:Ecco Reading(3181)
· Micron pushes TLC 128-Gbit NAND flash
Author:Ecco Reading(3662)
· Intel will stop supplying desktop motherboards
Author:Ecco Reading(5231)
· Processor market was expected to regain strength in 2013
Author:Ecco Reading(3248)
· It was reported that TSMC sales fall steeply
Author:Ecco Reading(3390)
· Cisco, NXP work with auto wireless startup
Author:Ecco Reading(3530)
· Micron was impacted by manufacturing glitch
Author:Ecco Reading(3935)
· China can make 22-nm transistor by themselves
Author:Ecco Reading(3707)
· Chip market rebound is coming, according to survey
Author:Ecco Reading(3677)
· Sony, Toshiba will spend more on chips, iSuppli reports
Author:Ecco Reading(3714)
· Qualcomm becomes the 13th company to join NFC Forum board
Author:Ecco Reading(6028)
· TSMC increases building work for FinFET fab
Author:Ecco Reading(3692)
· TI plans to cut 1,700 jobs in OMAP shift
Author:Ecco Reading(4478)